Sartorius acquires Albumedix: World leader in advanced therapies based on recombinant albumin
Sartorius acquires all shares of Albumedix Ltd. biotech company based in the United Kingdom. Albumedix Ltd. is a leader in the field of solutions based on recombinant albumin. Recombinant human albumin is a key component in the production of innovative biopharmaceuticals, especially for cell therapies, viral therapies and vaccines.
The Sartorius Group, through its French subgroup Sartorius Stedim Biotech, concluded an agreement on the acquisition of the biotechnology company Albumedix Ltd. from private investors. Based in Nottingham, UK, the company provides the best solutions based on recombinant albumin. Recombinant human albumin is an important component for the biopharmaceutical industry required for various applications, for example as an animal-free additive to cell culture media and for the stabilization of vaccines and viral therapies.
Company Albumedix Ltd. was founded in 1984 and has more than 100 employees. It is expected to generate revenue of approximately £33 million in 2022 with a significant double-digit EBITDA margin. The agreed purchase price amounts to approximately £415 million.
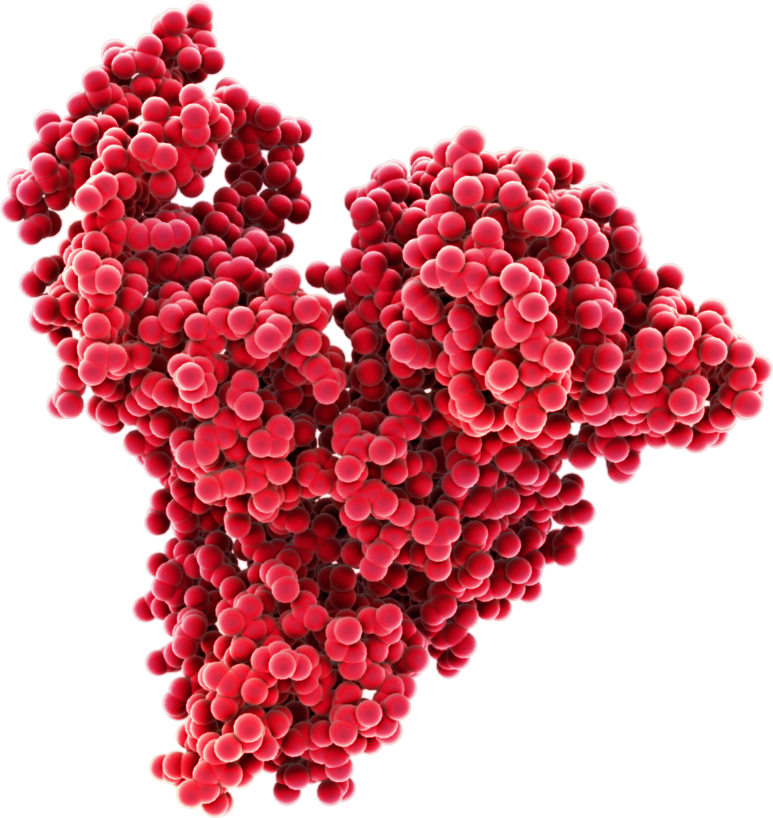
Human serum albumin (HSA) is the most abundant protein in human blood, constituting approximately half of the total plasma proteins. Albumin is responsible for maintaining oncotic pressure, plasma pH and distribution of a variety of molecules, making it a very well-studied protein. The structure of albumin gives it excellent stability, making it resilient to environmental stress both outside and inside the body.
Image source: Albumedix Ltd.
Albumedix will be an important addition to Sartorius’ advanced therapy solutions, particularly regarding our cell culture media business, as it will enable us to strengthen our position as a relevant supplier of innovative chemically defined media and critical ancillary materials.
This market offers high growth potential due to the increasing regulatory requirements as well as rising demand for the use of recombinant human albumin in near-patient applications. Albumedix will also add important formulation excipients to our vaccine production solutions, allowing us to expand our existing customer relationships and forge new ones.
René Fáber, member of the Executive Board for the Bioprocess Solutions Division of Sartorius
Albumedix will be part of the Bioprocess Solutions Division, and the existing 72,000-square-foot site in Nottingham will be established as a center of excellence for innovation and GMP-compliant production of critical raw materials.
For more information, read the official Sartorius Press Release.