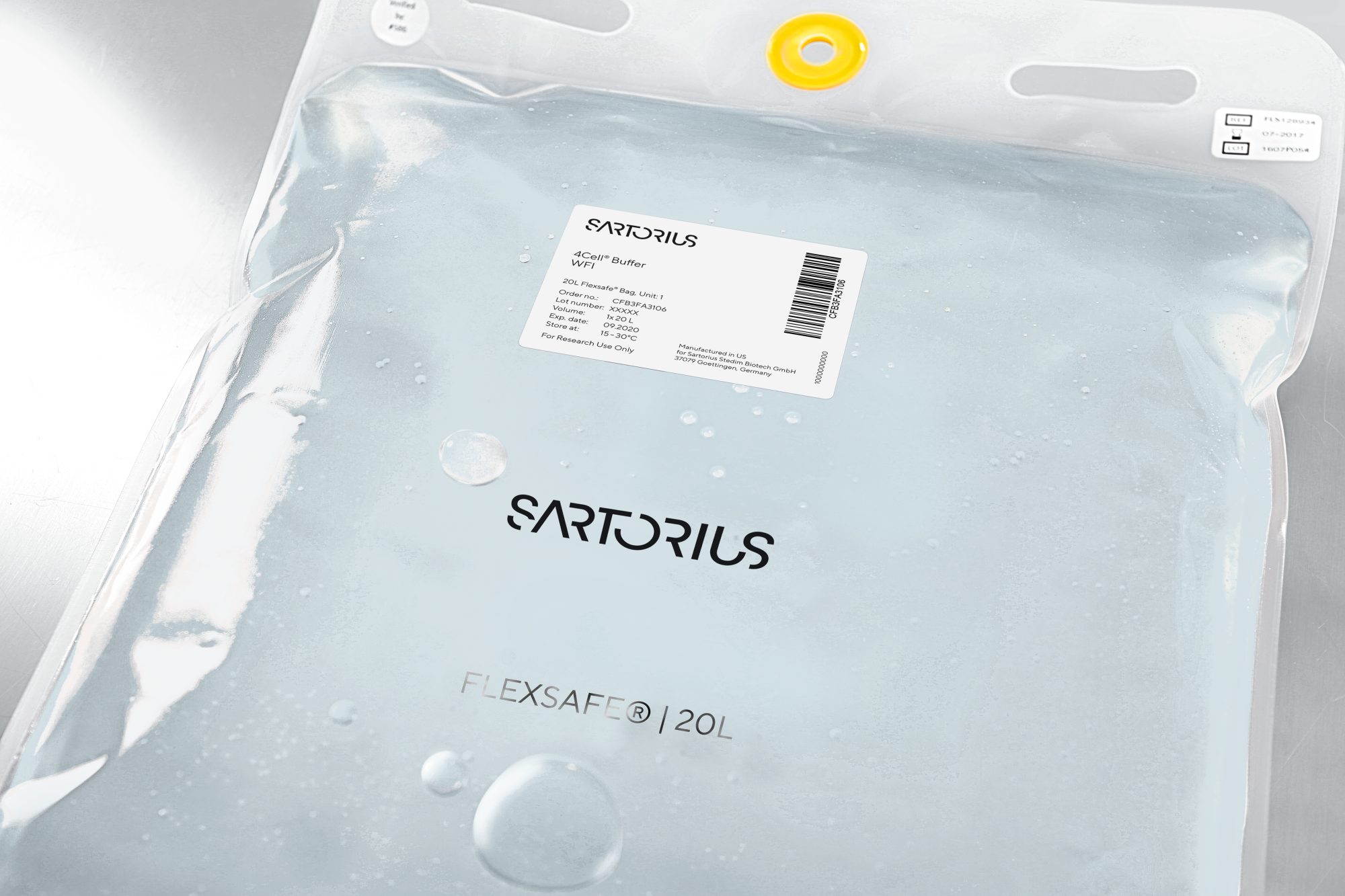
4Cell® Water for Injections
Our sterile filtered water for injections (WFI) is used in laboratory applications and production. It is manufactured and tested in accordance with strict industry standards. Meets or exceeds the requirements of the USP and EP. This product was produced aseptically under cGMP 21CFR820 conditions by a validated sterile filtration method. Our sterile filtered water for injections (WFI) can be used to prepare media and buffers, hydrate, clean, wash, rinse filters or equipment.
Price: On request
Water for Injection (WFI) — Sterile Water for Laboratory and Pharmaceutical Use
4Cell® Water for Injection (WFI-Quality Water) is aseptically processed water that meets current USP and EP specifications. It is intended for laboratory applications and bioprocess manufacturing — not for parenteral administration or IVD use.
What is water for injection and what is it used for?
Water for Injection (WFI) is highly purified water used in the pharmaceutical and biotechnology industries. It undergoes ultrafiltration, reverse osmosis, deionization, distillation, and sterile filtration.
Main applications:
• Preparation of media and buffers for bioprocessing
• Cell culture hydration
• Rinsing of laboratory equipment and filters
• GMP manufacturing of biologics
• Process development and scale-up phases.
Benefits of 4Cell® Water for Injection
- Highest quality standards - manufactured according to 21CFR820 guidelines
- Fast implementation - Flexsafe® bags applicable at all process stages
- Regulatory compliance - released according to EP and USP specifications
- Cost savings - no need to install your own WFI system
- Scalability - 20 L and 200 L packaging for instant capacity increase
